
May 2004, last revised Jan 2019
Part 2
4. The fossil record
5. Common descent and common design
Scientists often distinguish five kingdoms of nature: prokaryotes (archaea/bacteria), protists (microorganisms with cell nuclei), plants, fungi, and animals. Each kingdom is divided into phyla, which are in turn divided into classes, orders, families, genera, and species. Well-known animal phyla include: cnidarians (e.g. corals, jellyfish), molluscs (e.g. clams, snails, squid), echinoderms (e.g. starfishes, sea urchins), sponges, annelids (e.g. earthworms, leeches), arthropods (e.g. trilobites, insects, lobsters), chordates (all vertebrates, including reptiles, fish, mammals).
Humans are assigned to the kingdom Animalia, the phylum Chordata, the subphylum Vertebrata, the class Mammalia, the order Primates, the family Hominidae, the genus Homo, and the species sapiens.
Darwin envisaged one species slowly changing into a new one, which then changed into another one, until finally not just new species belonging to the same genus were produced, but new genera, families, orders, classes, phyla, and ultimately kingdoms of organisms evolved. The fossil record demolishes this model of ‘phyletic gradualism’.
Stephen J. Gould said that ‘The fossil record with its abrupt transitions offers no support for gradual change’ and ‘The extreme rarity of transitional forms in the fossil record persists as the trade secret of paleontology’.1 In his view, Darwin’s rationalization that the gaps were due to the ‘extreme imperfection’ of the fossil record is by now utterly untenable. Gould also stated that preserved transitions are ‘often found in the fossil record’ but ‘not common’, and added: ‘it is infuriating to be quoted again and again by creationists ... as admitting that the fossil record includes no transitional forms. Transitional forms are generally lacking at the species level, but they are abundant between larger groups.’2 Gould therefore made conflicting noises about the prevalence of transitional sequences. The fact that some evolutionists classify many species as ‘transitional’ (see below) and are able to assemble a few more or less progressive fossil sequences does not alter the fact that there are enormous gaps in the fossil record. As Eugene Koonin puts it:
Major transitions in biological evolution show the same pattern of sudden emergence of diverse forms at a new level of complexity. The relationships between major groups within an emergent new class of biological entities are hard to decipher and do not seem to fit the tree pattern that, following Darwin’s original proposal, remains the dominant description of biological evolution. The cases in point include the origin of complex RNA molecules and protein folds; major groups of viruses; archaea and bacteria, and the principal lineages within each of these prokaryotic domains; eukaryotic supergroups; and animal phyla. In each of these pivotal nexuses in life’s history, the principal ‘types’ seem to appear rapidly and fully equipped with the signature features of the respective new level of biological organization. No intermediate ‘grades’ or intermediate forms between different types are detectable.3

Fig. 4.1. The traditional ‘tree of life’ presupposes a single common ancestor. Nowadays, Darwinists believe that life may have arisen multiple times, and that evolution has produced a bush or orchard rather than a tree.4
It is estimated that 20 to 30 million species are alive today, though fewer than 2 million have been documented in the professional literature. Over 99% of species that have ever lived are believed by Darwinists to be extinct – some 200 million of them. But only about 150,000 species of extinct organisms have so far been catalogued on the basis of fossil evidence.5 Since 90 to 99% of the sedimentary rocks in which fossils might once have been preserved have been destroyed by erosion, the fossil record is certainly incomplete. Nevertheless, the fossil species already found offer a good random sampling of all the creatures that have existed. As far as terrestrial vertebrates are concerned, about 98% of living orders and 79% of living families have been found in fossilized form.6 But we do not see species slowly changing into completely different species as we trace them back in time.
If phyletic gradualism were true, species should be undergoing constant modifications, and we would expect to find fossils of many of the ‘inconceivably great’ number of transitional forms that Darwin admitted his theory required. If fish evolved into amphibians, for instance, we would expect to find intermediate forms showing the gradual transition of fins into legs and feet, and of fish scales into amphibian skin (which later transformed itself into reptilian scales). Since the transition would have required many millions of years, during which many hundreds of millions of transitional forms must have lived and died, many of them should have been discovered in the fossil record. Similarly, if reptiles evolved into birds, we would expect to find fossils showing the gradual transition of the forelimbs of the ancestral reptile into the wings of a bird, and the gradual transition of scales into feathers, hind feet into perching feet, the reptilian skull into the birdlike skull, etc. But as Gould says:
the absence of fossil evidence for intermediary stages between major transitions in organic design, indeed our inability, even in our imagination, to construct functional intermediates in many cases, has been a persistent and nagging problem for gradualistic accounts of evolution.7
Gould says that the history of most fossil species includes two features particularly inconsistent with gradualism:
1. Stasis. Most species exhibit no directional change during their tenure on earth. They appear in the fossil record looking much the same as when they disappear; morphological change is usually limited and directionless.
2. Sudden appearance. In any local area, a species does not arise gradually by the steady transformation of its ancestors; it appears all at once and ‘fully formed.’8

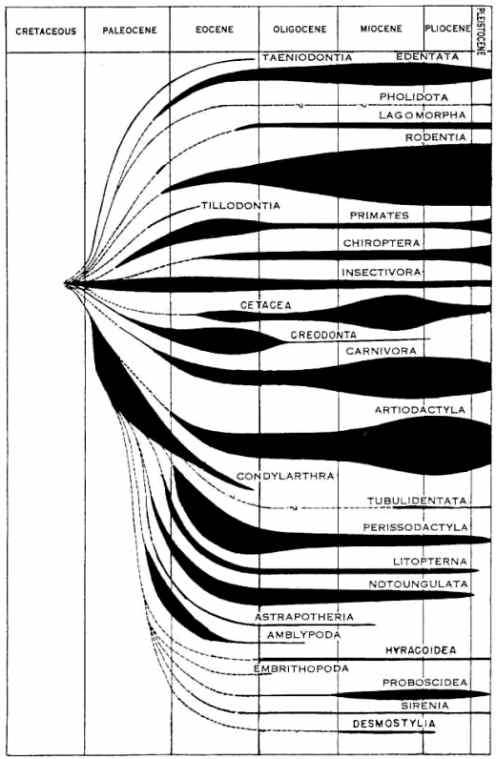
Fig. 4.2. In this diagram of dinosaur ancestry all the blue lines and dashed lines refer to inferred fossils, i.e. fossils that have never been found.9 In other words, all known dinosaur species represent only the twigs on the supposed evolutionary tree or bush; Darwinists cannot offer a single compelling example of an ancestral sequence for the dinosaurs.
According to Stephen Stanley, ‘The fossil record does not convincingly document a single transition from one species to another.’ Ernst Mayr says: ‘There is no clear evidence for any change of a species into a different genus, or for the “gradual emergence” of any evolutionary novelty.’10 And Eldredge writes:
Most families, orders, classes, and phyla appear rather suddenly in the fossil record, often without anatomically intermediate forms smoothly interlinking evolutionarily derived descendant taxa [groups of organisms] with their presumed ancestors.11
It is highly significant that the gaps in the fossil record become larger, the higher the taxonomic level, even though according to the Darwinian theory there must have been many times more transitional forms at higher levels. Horses, for example, belong to the family Equidae (order Perissodactyla), while bears belong to the family Ursidae (order Carnivora). According to standard Darwinism, the divergence between orders, e.g. between bears and horses, should have taken far longer and left behind more fossils than subsequent minor changes among bears or horses. But as Hoyle and Wickramasinghe point out, the evidence is the other way round, and this is the case for all classes of animals, not just mammals.
[T]he small divergences are there, the big are absent. We do not see part-bear, part-horse. Even within a single order, families remain stubbornly distinct from one another. For instance, the order Carnivora includes cats and dogs, and it is obvious that we see no evidence whatsoever of part-cat, part-dog.12
As Jeffrey Schwartz says:
the truth of the matter is that we are still in the dark about the origin of most major groups of organisms. They appear in the fossil record as Athena did from the head of Zeus – full-blown and raring to go ...13
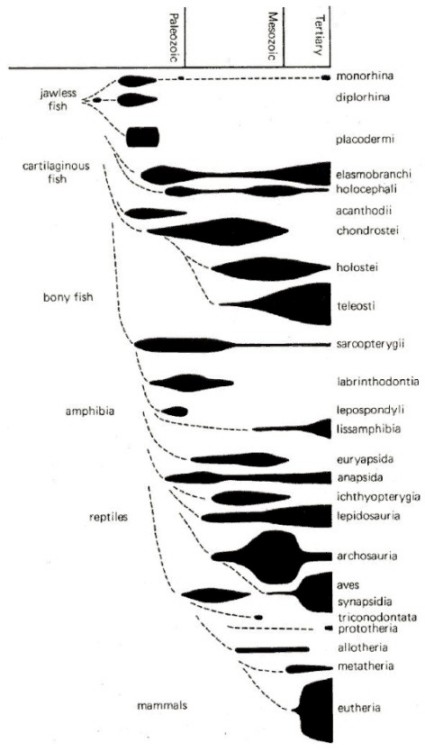
Fig. 4.3. The fossil record for the main vertebrate
groups (above) and orders of mammals (below).
The widths of the solid
areas indicate the changing numbers of species, and the dotted lines
represent hypothetical lineages, or missing evolutionary
links.14
In 1977 Gould and Eldredge reviewed cases of supposed phyletic gradualism, including several standard examples taught to students for decades, and found them unsatisfactory or downright false.15 As Stanley says, ‘The known fossil record fails to document a single example of phyletic (gradual) evolution accomplishing a major morphologic transition and hence offers no evidence that the gradualistic model can be valid.’16
Gould acknowledges that the small gradual changes observed in the fossil record are so tiny that they cannot reasonably be extrapolated into large-scale evolution:
[W]ell-represented species are usually stable throughout their temporal range, or alter so little and in such superficial ways (usually in size alone), that an extrapolation of observed change into longer periods of geological time could not possibly yield the extensive modifications that mark general pathways of evolution in larger groups. Most of the time, when the evidence is best, nothing much happens to most species.17
 As
Peter Williamson says, ‘conventional
neo-Darwinism ... has failed to predict the widespread long-term morphological
stasis now recognized as one of the most striking aspects of the fossil record’.18 On average, plant or animal species tend to go extinct
after about 4 million years, but some creatures have lasted far longer without
undergoing any marked change –
though random genetic drift would be expected to alter appearances even without
adaptive pressures. 90 kinds of cyanobacteria (blue-green algae), for instance,
have survived with little change for a billion years. The trilobites (fossil
shown right) burst onto the scene in the early Cambrian but then changed little
for 300 million years. Marine shellfish have existed unchanged for 10 to 14 million
years. The new species of seabed Foraminifera that appeared in the early Cenozoic
were typically able to survive, unaltered, for at least a further 20 million
years.
As
Peter Williamson says, ‘conventional
neo-Darwinism ... has failed to predict the widespread long-term morphological
stasis now recognized as one of the most striking aspects of the fossil record’.18 On average, plant or animal species tend to go extinct
after about 4 million years, but some creatures have lasted far longer without
undergoing any marked change –
though random genetic drift would be expected to alter appearances even without
adaptive pressures. 90 kinds of cyanobacteria (blue-green algae), for instance,
have survived with little change for a billion years. The trilobites (fossil
shown right) burst onto the scene in the early Cambrian but then changed little
for 300 million years. Marine shellfish have existed unchanged for 10 to 14 million
years. The new species of seabed Foraminifera that appeared in the early Cenozoic
were typically able to survive, unaltered, for at least a further 20 million
years.
Some extant vertebrates have never shown any evolutionary changes during a species lifetime of at least 100 million years. The common freshwater ‘fairy shrimp’ Triops differs from specimens preserved in rocks 180-200 million years old only in having grown slightly bigger since that time. The coelacanth and lungfishes appear to be wholly unchanged even after 300 million years – twice as long as the duration of the age of dinosaurs. The lamp shell Lingula is a ‘living fossil’ that has remained essentially unchanged for 450 million years. And the tuatara lizard has shown little change for nearly 200 million years since the early Mesozoic. 84% of insect families alive today were alive 100 million years ago but have resisted evolutionary change. The now-living mammals of Europe seem to have remained unchanged for the past million years.
Began (years BP) |
|
| Phanerozoic eon | |
| Cenozoic era | |
| Quaternary period: | |
| Holocene epoch | 11,700 |
| Pleistocene | 2,588,000 |
| Tertiary period: | |
| Pliocene epoch | 5,333,000 |
| Miocene | 23,030,000 |
| Oligocene | 33,900,000 |
| Eocene | 56,000,000 |
| Palaeocene | 66,000,000 |
| Mesozoic era | |
| Cretaceous | 145,000,000 |
| Jurassic | 201,300,000 |
| Triassic | 252,170,000 |
| Palaeozoic era | |
| Permian | 298,900,000 |
| Carboniferous | 358,900,000 |
| Devonian | 419,200,000 |
| Silurian | 443,400,000 |
| Ordovician | 485,400,000 |
| Cambrian | 541,000,000 |
| Proterozoic eon | 2,500,000,000 |
| Archean eon | 4,000,000,000 |
| Hadean eon |
4,600,000,000 |
Fig. 4.4. The scientific geological timescale (for corresponding theosophical dates, see section 8).
References
Although the myriads of transitional forms that Darwin expected to turn up have failed to do so, there are still numerous species that many Darwinists classify as transitional. Any species which combines features from two more or less successive groups of organisms is automatically assumed to belong to a series of intermediate forms linking the two groups. The vast majority of ‘transitional’ species are regarded as possessing too many specialized features to be ‘true’ ancestral species and are classed as ‘close relatives’ of the real ancestors, which are still missing. What is never found in the fossil record is creatures with clearly half-finished features or mutant creatures with nonfunctional features, yet random mutations should have produced countless failures of this kind, which would then have been eliminated by natural selection.
Over 95% of all fossils are marine invertebrates, mostly shellfish. Of the remaining 5%, most are plants. Less than 1% of all fossils are fish, and even fewer are land animals. The various types of invertebrates all appeared abruptly in the fossil record; there is no hint of how they originated from unicellular life, and there are no signs of gradual evolution of one into another. The fossils are often of entire specimens, which makes it difficult for evolutionists to speculate about ‘transitionals’. Fossils of plants, algae and fish also show no convincing evidence of gradual evolution. Most of the transitional fossils presented by Darwinists are land-dwelling vertebrates. Fossil species are often represented by a bone or less; this means that interpretations are very subjective, and there is serious disagreement among palaeontologists about which specimens qualify as transitional, and which supposed transitional forms fit into which lineages and where.1

Fig. 4.5. Late Devonian lobe-finned fish are claimed to have evolved into amphibious tetrapods.2
Darwinists argue that tetrapods (four-limbed animals such as amphibians, reptiles, birds and mammals) evolved from the lobe-finned fishes (Rhipidistia) in the Devonian period. In 1938 fishermen in the Indian Ocean hauled to the surface a lobe-finned coelacanth (pronounced: SEE-la-kanth), a living relative of the ancient lobe-finned fishes. The coelacanths appeared about 400 million years ago and were thought to have gone extinct 100 million years ago. On the basis of fossil evidence, it had been touted as a missing link between fishes and amphibians, but these hopes were dashed once the soft anatomy of a living specimen could be examined. Scientists had envisioned coelacanths dragging themselves along the ocean floor with their lobed limblike fins, but it turned out that they swim rather than crawl. This shows how difficult it is to draw conclusions about the overall biology of organisms from their skeletal remains alone. The coelacanth is just another peripheral twig on the presumed tree of life. The lungfish, another type of lobed-fin fish, has fins, gills and an intestine containing a spiral valve like any fish, but lungs, heart and a larval stage like an amphibian. But although it has a mixture of fish and amphibian traits, the individual characteristics are not in any realistic sense transitional between the two types, and it is no longer seen as ancestral to tetrapods.


Fig. 4.6. Ichthyostega, one of the first tetrapods, beneath a presumed fish ancestor (Eusthenopteron).3 Ichthyostega, a labyrinthodont, combines a fishlike tail and gills with an amphibian skull and limbs but, like Acanthostega, was predominantly aquatic. Acanthostega had eight fingers and Ichthyostega had seven toes.
The next candidate for the missing link between marine and terrestrial life was Eusthenopteron, a lobe-finned fish sharing various features with the earliest known tetrapods. It used to be depicted as venturing onto land, but is now widely believed to have been strictly aquatic – a shallow-water predator. More recently, elpistostegids such as Panderichthys and Tiktaalik (fig. 4.5) have been loudly touted as ancestors, or near-ancestors, of the tetrapods, and have been dubbed ‘fishapods’. These creatures had paired fins, rather than true arms and legs, and were capable of very limited crawling. After its discovery in 2006, Tiktaalik was hailed as the closest ancestor of the tetrapods, because its pectoral (front) fins had a wrist-like feature with radial bones. However, some scientists have decided that the fins of Panderichthys are more tetrapod-like, even though this fish had previously been said to shed little light on the origin of major features of the skull, limbs and axial skeleton of early tetrapods. Panderichthys had ‘front-wheel drive’, meaning that its front fins were bigger and more powerful than its rear fins. The early tetrapods, however, were ‘rear-wheel drive’. Darwinists therefore expected to find a fossil illustrating the emergence of hind-limb-powered propulsion in the interval between Panderichthys and Acanthostega. Tiktaalik, however, was more of a ‘front-wheel drive’ animal than Panderichthys.4 A further problem is that the oldest elpistostegid fossils are 386 million years old, but the discovery of earlier footprints and tracks has pushed back the emergence of the first land animals to 395 million years ago.5
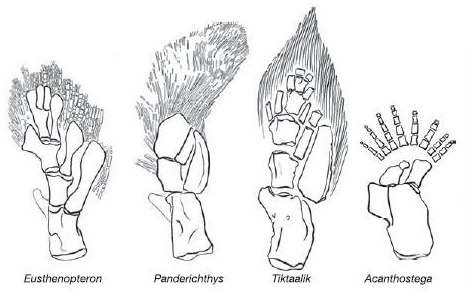
Fig. 4.7. The ray-finned fish fins of Eusthenopteron, Panderichthys and Tiktaalik compared with the tetrapod limb of Acanthostega. The radial bones in fins allegedly developed into the digits of tetrapod limbs, but there is still a huge gap to be bridged.6
There is certainly a degree of continuity between the lobe-finned fish and the first tetrapods. For instance, both Acanthostega and Ichthyostega ‘retain a sort of fish shape, they apparently retained internal gills, they retain a lateral sensory line on the body (as in fish) and they have a sort of tail fin – albeit of a different shape’.7 But there are still huge discontinuities regarding the supposed transformation of fins into limbs and the development of terrestrial locomotion. For instance, the fossil record fails to document how a functional pelvic or shoulder girdle slowly evolved in the transition from fish to early amphibians. And the chips of cartilage or bone present in the fins of certain fish are loosely embedded in muscle and not connected to the backbone at all. Devonian tetrapods show a mosaic of terrestrial and aquatic adaptations, but it is difficult to see them as ‘true’ ancestors of later creatures. Ichthyostega, for example, is described as ‘a very strange animal’, ‘a curious mixture of features, some of them primitive but some of them specialized and unique’; the shoulder girdles of the early tetrapods are ‘not obviously halfway in structure between those of fishes and those of later tetrapods’ but possess ‘some unique and some unexpected features’.8
Seymouria, a reptile-like labyrinthodont from the early Permian, is a commonly touted intermediate between amphibians and reptiles. In terms of purely skeletal characteristics it would appear to be a convincing intermediate, but there is a serious problem. The major difference between amphibians and reptiles lies in their reproductive systems. Amphibians lay their eggs in water and their larvae undergo a complex metamorphosis (like a tadpole) before reaching the adult stage. Reptiles develop inside a hard shell-encased egg and are perfect replicas of the adult on first emerging, and the problems of envisaging the gradual evolution of the reptilian egg are immense. But fossil evidence suggests that Seymouria was wholly amphibian in its reproductive system. A further difficulty is that Seymouria appears in the fossil record about 30 million years later than the earliest true reptiles, Hylonomus and Paleothyris.9
The small caterpillar-like organism Peripatus is sometimes put forward as an intermediate between the annelid worms and the arthropods. But once again, its organ systems are not strictly transitional between the two groups. For example, its circulatory and respiratory systems are typically arthropod in their basic design, while its nervous and excretory systems are typical of those seen in many annelid worms. Peripatus is really a mosaic of characteristics drawn from two distinct groups, as is the lungfish, and also the duckbill platypus (a monotreme or egg-laying mammal), which is reptilian in so far as it lays eggs, but entirely mammalian in its possession of hair, mammary glands and three ear bones. As Denton says:
they provide little evidence for believing that one type of organism was ever gradually converted into another. ... Between lungfish and amphibia, between monotremes and reptiles and between Peripatus and arthropods, there are tremendous gaps unbridged by any transitional forms.10
The transition from mammal-like reptiles to mammals in the Triassic is usually claimed to be best documented case of macroevolution in the fossil record. However the ‘sequence’ from pelycosaurs to therapsids to mammals contains many gaps, which neo-Darwinists assume will one day be filled by new fossil discoveries. Mammalian traits sometimes appear, disappear, and then reappear, they sometimes appear out of sequence, and they often emerge independently – and supposedly by chance – in different lineages.11 Some researchers argue that, on present fossil evidence, it is unlikely that pelycosaurs evolved into therapsids, but one of the six orders of therapsids (the cynodonts) did become increasingly mammal-like, and one branch of cynodonts may have evolved into morganucodonts (an extinct order of primitive mammaliaformes). However, no Mesozoic group can be identified as the ancestors of modern mammals.12

Fig. 4.8. Above: The whale
and some of its alleged ancestors, drawn to scale.
Below: Two supposed
intermediate species.13
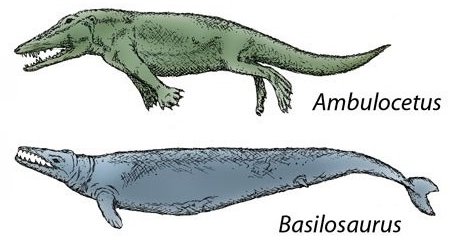
Whales – marine mammals with a carnivorous diet – appeared abruptly in the early Cenozoic. Darwin hypothesized that whales evolved from bears. Later evolutionists have proposed several other hypothetical ancestors: mesonychids (wolf-like carnivores), the hyena (Pachyaena), Sinonyx (a cat-like creature), herbivorous hippopotami, and Indohyus, a herbivorous deer-like creature. The transformation would have required radical changes in reproduction, respiration and locomotion, but there is no smooth fossil sequence showing that it ever occurred. Proposed intermediate species include: Pakicetus, a land dweller partially adapted to aquatic life, usually classified as a primitive whale based on certain features of its teeth and ears; Ambulocetus, a 3-metre-long, amphibious crocodile-like creature; and Basilosaurus, a 20-metre-long, eel-like creature whose features, according to more cautious evolutionists, preclude it from being a true whale ancestor.14 These forms each have their own distinguishing characteristics, which they would have to lose in order to be considered direct ancestors or descendants of one another.15
Birds are nowadays classed as a type of dinosaur; most scientists believe they evolved in the Jurassic period from a group of feathered dinosaurs known as theropods. However, dinosaurs were already very specialized, and due to this and other problems some scientists believe birds evolved from some other group of extinct reptiles.16 The ancient bird-dinosaur Archaeopteryx from the late Jurassic is widely regarded as a close relative of the ancestor of modern birds. The specimens of this primitive bird range from the size of a blue jay to that of a large chicken. Archaeopteryx possessed reptilian features such as teeth, a long lizard-like tail, and claws on its wings. However, it also possessed bird-like characteristics, such as a perching foot, a wishbone, and fully formed wings covered not with ‘primitive’ feathers but with modern flight feathers. Just how well it could fly is disputed. Archaeopteryx hints at a reptilian ancestry, but it is not led up to by a series of transitional forms from an ordinary terrestrial reptile through a number of gliding types with increasingly developed feathers until the full avian condition is reached. Furthermore, fossil tracks of birds have been found in far older strata, dated at 212 million years ago (late Triassic). And controversial fossils of a more modern-looking bird named Protoavis have also been found in strata of late Triassic age.17

Fig. 4.9. The feather is both extremely light and structurally strong – an engineering marvel.18 A single pigeon feather may have several hundred thousand barbules and millions of hooklets (hamuli).
There are several instances in the fossil record where a relatively minor morphological transformation can be traced through a series of fossil forms. The best-known case is that of the horse. The series starts with the original dog-sized horse, Eohippus (Hyracotherium), which lived about 50 million years ago and had four toes on the fore limbs and three on the hind limbs. It then passed through three-toed varieties, and ended with the modern one-toed Equus. However, the evolution of the horse is now admitted to have been much more complicated than originally assumed. Many species coexisted with ‘ancestor’ species, and instead of a progressive change in traits, several reversals took place. Many species appeared abruptly and remained unchanged throughout their lifetimes, species appeared that are entirely inconsistent with the supposed overall ‘trend’, and three-toed horses and one-toed horses commonly coexisted in North America.19
The differences between Eohippus and the modern horse are relatively trivial, yet the two forms are separated by 50 million years and at least 10 genera and a great number of species. The horse series therefore emphasizes just how vast the number of genera and species must have been if all the diverse forms of life on earth had really evolved in the gradual way that neo-Darwinism implies. There must have been innumerable transitional species linking such diverse forms as land mammals and whales or molluscs and arthropods. Yet they have barely left a trace of their existence in the fossil record. This seems to leave a saltational model as the only evolutionary explanation of the gaps.
References
The earliest forms of life are thought to have originated some 3.85 billion years ago. They were unicellular microorganisms – bacteria and archaea – composed of prokaryotic cells (cells without a nucleus). The more complicated eukaryotic (nucleated) cell appeared by about 2 billion years ago, and is found in the unicellular protozoans, algae and lower fungi. Its advent marks a major discontinuity in the sequence of living things.
Some prokaryotes were multicellular, but complex multicellular organisms evolved only among eukaryotes. The great radiation and diversification of the multicelled animals, or metazoans, began towards the end of the Precambrian, with the appearance of the Ediacaran (or Vendian) fauna. The radiation attained its climax in the succeeding ‘Cambrian explosion’ from about 530 to 520 million years ago. Just how one or more singled-celled organisms evolved into metazoans and what intermediates were involved is one of the great unsolved puzzles of evolution. There is a large gap between single-celled and multicelled animals, as there is no known animal with two, three, four, or even 20 cells. Moreover, not only has multicellularity evolved separately in the three great higher kingdoms of life (plants, fungi and animals), but it is thought to have arisen several times in each kingdom.
The earliest unequivocal evidence of metazoan life dates from about 600 million years ago. Most Ediacaran fauna appeared abruptly and fully formed about 575 million years ago, after a severe glaciation, and went extinct before the start of the Cambrian.1 They comprised a wide variety of soft-bodied, shallow-water marine invertebrates, similar to modern-day jellyfish, lichens, soft corals, sea anemones, sea pens, annelid worms, and seaweed, along with some organisms unlike any known today. Disc-shaped fossil impressions are commonly a few centimetres across, occasionally reaching 20 cm, while frond-shaped impressions can attain lengths of about 1 metre. There are also ‘trace fossils’, resembling tracks and burrows. The fauna are mostly variations on a single anatomical plan: a flattened form divided into sections that are matted or quilted together – a design no longer found today. Although originally regarded as precursors of some of the later, Cambrian creatures, it is now widely believed that most were unrelated to anything that came afterwards and were a failed experiment. However, metazoan animals of modern design, such as sponges, shared the earth with the Ediacaran fauna.



Fig. 4.10. Three Ediacaran fossils. Top to bottom: Dickinsonia costata, displaying the characteristic quilted appearance of Ediacaran fauna; opinions differ on whether it is an animal, fungus or something else; Charniodiscus arboreus, probably a stationary filter feeder, with a holdfast, stalk and frond; Spriggina floundersi (4 cm long): its affinity is currently unknown – it has been classified as an annelid worm, a frond and an arthropod.2
The first worldwide fauna of creatures with hard body parts (such as calcium carbonate shells) date from just before the start of the Cambrian to about 10 million years after. This ‘small shelly fauna’ includes spines, sclerites (armour plates), tubes, archaeocyathids (sponge-like animals) and small shells similar to those of brachiopods and snail-like molluscs. They are mostly 1 to 5 mm in length, and the bulk are fragments of the skeletons of larger organisms. These fossils may represent another failed experiment. The extinction of most of these forms about 530 million years ago was followed by the most dramatic phase of the Cambrian explosion.
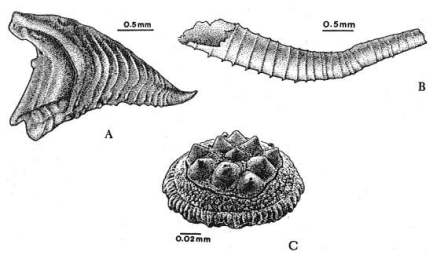 |
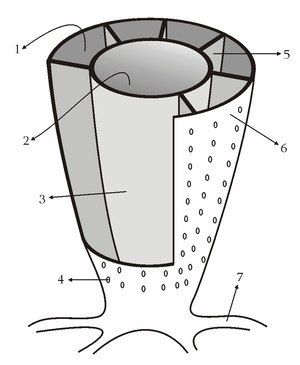
Fig. 4.11. Above: Representative organisms of the ‘small shelly fauna’. Below: The most characteristic and abundant of all these creatures are the archaeocyathids, the first reef-forming creatures. 1 = gap; 2 = central cavity; 3 = internal wall; 4 = pore (all walls have pores); 5 = septum; 6 = outer wall; 7 = holdfast.3 |
 |
The Cambrian explosion is one of evolution’s greatest mysteries.4 Charles Darwin considered this sudden appearance of many animal groups (in strata initially included in the Silurian period in his day) with no known ancestors to be the gravest challenge to his theory of evolution. Within no more than 10 million years, about 20 of the roughly 26 animal phyla present in the known fossil record made their first appearance; three appeared in the Precambrian, and three are currently thought to have appeared after the Cambrian, though it cannot be ruled out that all phyla had appeared by the end of the Cambrian or even the end of the early Cambrian.5 Some Cambrian fossils probably represent additional phyla that have since gone extinct.6 All the (non-algal) divisions (phyla) of plants are thought to be post-Cambrian.
There is disagreement about how many phyla are represented in Cambrian strata, and how to classify certain creatures, but there is no denying that the event represents an extraordinary profusion of new body plans. They include clams, snails, trilobites, brachiopods, worms, jellyfish, sea urchins, sea cucumbers, swimming crustaceans, sea lilies, and other complex invertebrates. Trilobites abound, and their eyes were at least as proficient as those possessed by any animal living today. Although the creatures differ drastically from one another, Darwinists like to believe that they all evolved from the same hypothetical common ancestor – probably a flatworm-like creature that lived several hundred million years earlier. If the Cambrian biota had arisen through step-by-step variations, there should be evidence of countless transitional forms over vast stretches of geologic time. But, as Stephen Meyer points out, ‘Instead of more and more species eventually leading to more genera, leading to more families, orders, classes, and phyla, the fossil record shows representatives of separate phyla appearing first followed by lower-level diversification on those basic themes.’7 The history of animal life has largely been a tale of endless variations on the basic body plans that emerged during the Cambrian.
| A | 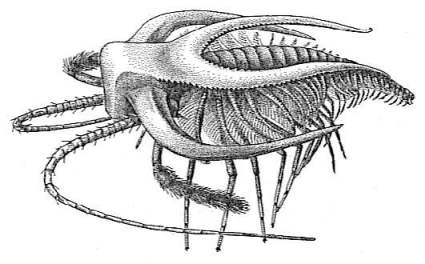 |
| B | 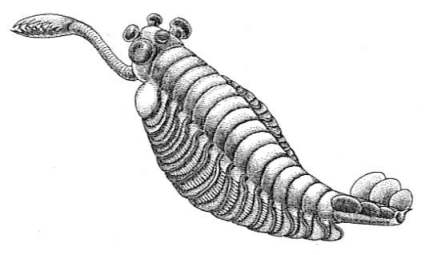 |
Fig. 4.12. Four arthropods from the Cambrian explosion. A. Marrella, ranging from 2.5 to 19 mm in length. B. Opabinia, 43 to 70 mm long, showing the frontal nozzle with terminal claw, five eyes on the head, and body sections with gills on top. C. Reconstruction of Sidneyia. D. Anomalocaris; this predator was up to 1 metre long.8 |
|
C |
 |
| D |  |
There have been several other notable radiations of new life forms since the Cambrian explosion. For instance, the Great Ordovician Biodiversification Event (GOBE) saw a staggering increase in marine biodiversity, largely within the phyla established during the Cambrian explosion. Diversity at order, family, genus and species level tripled during a period of 25 million years, making this event the most dramatic diversity explosion in the earth’s history.9
Land plants first appeared in the Ordovician and underwent a rapid radiation during the Devonian period, about 400 million years ago. The Devonian is also known as the ‘age of fishes’, due to the tremendous diversification of numerous extinct and modern major fish groups. The flowering plants (or angiosperms), a phylum that includes all the grasses, palms and all nonconiferous trees, originated some 140 million years ago during the Cretaceous period, and have diversified into about 250,000 different species. None of the defining characteristics of angiosperms, including the key novelties of the flower – sepals, petals, stamens, carpels – is found in any other group of plants, extant or fossil. From the outset they were already divided into different classes, many of which have persisted with little change up to the present day. Darwin called their sudden emergence ‘an abominable mystery’, and there is still no fossil sequence showing exactly how flowers evolved.
There are currently an estimated 6 to 10 million species of insects, which potentially represent over 90% of animal life forms on earth. Insects’ evolutionary ancestry is a puzzle; the oldest known fossil dates from the Devonian and was already fully capable of winged flight. There have been four main radiations of insects: beetles (~300 million years ago), flies (~250 million years ago), and moths and wasps (~150 million years ago).
Just as there have been major radiations of new organisms, so have there been several major extinctions and many minor ones. The severest mass extinction occurred at the Permian-Triassic boundary, some 252 million years ago, and wiped out up to 96% of all marine species and 70% of terrestrial vertebrate species. Other mass extinctions occurred at the end of the Ordovician, in the late Devonian, and at the end of the Triassic, about 450, 374, and 201 million years ago respectively.
Another mass extinction took place about 66 million years ago, at the end of the Cretaceous period and the beginning of the Palaeogene (the K-Pg extinction event); it wiped out three-quarters of all animal and plant species, including the dinosaurs. The most popular explanation is that the earth was struck by an asteroid or comet, generating a huge dust cloud which blocked out sunlight and led to the collapse of the food chain. However, the extinctions began hundreds of thousands of years before the K-Pg boundary, and some scientists believe that the main causes were a long period of intense global volcanism, related climatic changes, and changes in sea level or land elevation.10 This extinction was followed by the rapid diversification and rise to dominance of the mammals.
The advent of the modern mammals after the death of the dinosaurs should have left the best-preserved fossils of intermediate species. 66 million years ago, most mammals were small nocturnal tree-shrew-like animals, and roughly 10 million years later we find essentially modern whales, dolphins, rodents, marsupials, anteaters, horses, camels, elephants, bears, lions, bats, etc. All modern orders of mammals seem to have arisen independently and at about the same time. Not only is there no gradual sequence of intermediate species, but anyone who tries to imagine a sequence of viable intermediate animals between, for example, a tree-shrew and a bat – each of which is ‘better adapted’ than its predecessor – will very soon be convinced that such a sequence is inconceivable. Moreover, modern bats appeared twice in the early Cenozoic.
Taxonomy, or systematics, is the science of biological classification, and seeks to arrange plants and animals into hierarchies of superior and subordinate groups on the basis of the features they have in common. Branching diagrams (cladograms) are drawn showing the affinities between different species, and many taxonomists then interpret each node where a new branch begins as representing a hypothetical common ancestor. Alec Panchen says that common descent ‘seems so obviously the correct answer to the apparent relationships of classification, that any rejection of that explanation must surely be due to ignorance, stupidity or prejudice’.1 However, the father of modern taxonomy, the 18th-century botanist Carl Linnaeus, considered the ease with which plants and animals fell into an orderly groups-within-groups system of classification, or nested hierarchy, to be evidence for design. Richard Owen, one of Darwin’s great rivals, believed that the novel features defining different taxa or types of organisms were ‘primal patterns’ or archetypes that reflected the transcendent purpose of the divine mind, but that God had used natural laws to achieve his ends.2
There are over 100,000 taxa-defining novelties. The defining features of the class of mammals, for example, include hair, an enucleated red cell, a diaphragm, mammary glands, and a cerebral cortex consisting of six layers. These features are not possessed even in rudimentary form by any other vertebrate class. The fossil record fails to reveal a long series of adaptive transitional forms leading up to any taxa-defining features. In fact, in many cases there is no sign that they ever served any specific adaptive function or survival purpose.3 The curious geometric and numeric patterns of some of the features strengthens the impression that they are abstract, nonadaptive forms. In many species of flowers, for example, the number of petals corresponds to a Fibonacci number (see Patterns in nature).
The taxa-defining novelties have remained invariant in divergent evolutionary lines for tens or hundreds of millions of years. For instance, the wide range of vertebrate limbs (fins for swimming, hands for grasping, wings for flying) are all modifications of the same ground plan: the pentadactyl (five-digit) limb (fig. 5.1) – a basic pattern that has been conserved in all tetrapods for 400 million years. The centipede ground plan, which includes a poison claw and an odd (never even) number of segments, has remained unchanged for 420 million years. The body plan of adult beetles has remained essentially unchanged for 300 million years.4

Fig. 5.1. The pentadactyl limb. (ib.bioninja.com.au)
Biologist Rupert Riedl has said: ‘If every character were free to change in every direction, the living world would appear as a random chaotic mixture of patterns ...’5 However, the Darwinian idea that all living forms are part of an ever-mutating continuum is clearly a fiction. As Michael Denton points out, ‘all the evo-devo evidence suggests that there are deep and profound developmental constraints that work against such infinite pliability’.6 Ironically, it’s precisely the invariance of the taxa-defining features that allows descent with modification to be inferred. Denton agrees that cladograms can be interpreted as evidence for descent with modification, but insists that this must have involved sudden, saltational, nonrandom transitions, rather than gradual, random change (see section 6).

Fig. 5.2. A cladogram.
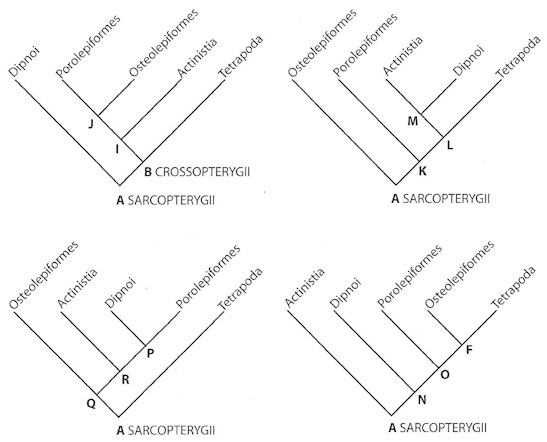
Fig. 5.3. Four examples of more than seven competing cladograms for the supposed transformation of fish into tetrapods.7
A group of dissident scientists, called ‘transformed cladists’ by their opponents, reject the hypothesis of common ancestry as unnecessary and see cladograms solely as a representation of a natural hierarchy of characteristics. Although they reject the a priori assumption of ancestor-descendant sequences (phylogeny), and express notable dissatisfaction with evolutionary theory and methods, most transformed cladists are in fact evolutionists, even though their peers regard them as traitors. They merely recognize that virtually all groups, living or extinct, are already too specialized to be reasonably called directly ‘ancestral’ to any other, and that none of the logically required truly ancestral forms are to be found in the fossil record. Only the outer twigs on the supposed evolutionary tree can be verified; the ancestral forms constituting its trunk and boughs are missing. As Gareth Nelson and Norman Platnick wrote in 1984: ‘We believe that Darwinism is a theory that has been put to the test in biological systematics, and has been found false.’8
Since the fossil record has not provided any substantial evidence of the evolutionary tree of descent that Darwinists expected to find, they now tend to speak of a labyrinthine ‘bush’. They acknowledge, however, that it is often difficult to judge where any given fossil falls among the many branches of the tree or bush. Robert Wesson writes:
Charts depicting ancestries through the ages are sometimes fudged by drawing connections where they are assumed; the more honest ones have dotted lines.
The gaps in the record are real ... The absence of a record of any important branching is quite phenomenal. Species are usually static, or nearly so, for long periods, species seldom and genera never show evolution into new species or genera but replacement of one by another, and change is more or less abrupt.9
And Ernst Mayr says:
It comes as rather a surprise to most nontaxonomists how uncertain our understanding of degrees of relationship among organisms still is today. For instance, it is still unknown for most orders of birds which other order is a given order’s nearest relative. The same is true for many mammalian families and genera ...Yet these uncertainties in the classification of higher vertebrates are very minor compared to those of the invertebrates, the lower plants, and most of all, the prokaryotes and viruses.10
David Raup points out that many scientists think the fossil record is far more Darwinian than it really is due to oversimplified textbooks, semipopular articles, etc. plus wishful thinking; ‘some pure fantasy has crept into textbooks,’ he says. Various ‘tricks’ are used to strengthen the impression of Darwinian descent. For instance, some authors display a series of fossils which show a progression in morphology, but which are not chronologically successive, and therefore cannot be evolutionary sequences. Alternatively, a chronologically successive series of teeth, jaw bones, etc. may be displayed as an evolutionary sequence, even though the author may know that the body parts are from organisms that could not reasonably have formed a lineage.11
References
Similarities in the structure, physiology or development of different species are said to be homologous if they are attributable to descent from a common ancestor. For instance, the forelimbs of humans, whales, dogs and bats are regarded as homologous, i.e. derived from an ancestor with similarly arranged forelimbs. Corresponding features with similar functions that are not thought to have originated by common descent are said to be analogous (or homoplasious). Examples are the wings of birds and flies, which are believed to have developed independently. Darwinists define homology in terms of common ancestry, but also cite it as evidence for common ancestry – a circular argument.
‘Homologous’ structures are supposed to have initially originated by the random accumulation of tiny advantageous mutations, and then to have been inherited by descendant species and further adapted, thanks to natural selection of further random mutations. ‘Analogous’ structures, on the other hand, are supposed to have arisen by random mutations several times and entirely independently – this is called convergent or parallel evolution. Parallel evolution refers to the appearance of similar patterns in more or less closely related plant and animal species, while convergent evolution refers to the appearance of striking similarities among organisms only very distantly related, but the boundary between the two terms is blurred.
Convergent evolution demonstrates that similarity does not always imply homology, i.e. inheritance from a common ancestor. There are many cases where similar features once classed as homologous have later been reclassified as analogous. Moreover, traits controlled by identical genes are not necessarily homologous, and homologous structures need not be controlled by identical genes. Regulatory genes that are considered homologous may be dedicated to non-homologous morphology. There are many examples where homologous structures develop via completely different embryological routes. For instance, the alimentary canal is formed from the roof of the embryonic gut cavity in sharks, from the floor in the lamprey, from the roof and floor in frogs, and from the lower layer of the blastoderm in birds and reptiles.1
There are hosts of convergences in the plant world. Very similar leaf patterns, for example, have appeared again and again in separate genera and families. Green plants depend for their survival on photosynthesis, whereby sunlight is used to convert water and carbon dioxide into energy-rich carbohydrates. 3% of plants use C4 photosynthesis, in which CO2 is first fixed into a four-carbon acid with the aid of an enzyme called PEPC. These acids then diffuse to the cells in an airtight structure known as the bundle sheath, where they are broken down into CO2 molecules, after which photosynthesis proceeds as normal. This highly complex and efficient process allows plants to grow faster and use less water. According to Williams et al., ‘C4 photosynthesis has independently evolved from the ancestral C3 pathway in at least 60 plant lineages, but, as with other complex traits, how it evolved is unclear’.2



Fig. 5.4. Three species of South American butterflies which closely mimic each other, even though they belong to distinct families: Melinaea lilis imitata, Helinconius ismenius telchinia, Dismorphia amphione praxinoe.3 Many close similarities are found in the wing colouration patterns of butterflies, both within and between families.

Fig. 5.5. Convergent evolution of the raptorial foreleg of the praying mantis and an insect known as Mantispa. It is derived from a generalized insect leg, modified for catching and grasping prey. It also evolved independently in a third group of insects, the rhachiberothidids.4
A striking example of convergent evolution is provided by the two main branches of the mammals, the placentals and marsupials, which have supposedly followed independent evolutionary pathways, after splitting off from some primitive mammalian common ancestor in the late Cretaceous. (Placentals bear their young fully developed, while marsupials give birth prematurely and nurture their young in a pouch.) The marsupials of Australia have evolved in isolation from placental mammals elsewhere yet have given rise to a whole range of similar forms: pouched versions of anteaters, moles, flying squirrels, cats, wolves, etc. Much the same phenomenon occurred in South America, where marsupials independently gave rise to a range of parallel forms.

Fig. 5.6. Examples of convergence: placental and marsupial mouse, placental wolf and marsupial Tasmanian wolf, marsupial flying phalanger and placental flying squirrel.

Fig. 5.7. Convergence in the sabre-tooth: drawing by Carl Buell of the placental Smilodon (top) and the marsupial Thylacosmilus.5
The eye has appeared many times in unrelated groups of animals. There are two main types of eye: the compound eye found in arthropods, and the camera eye, both of which first appeared in the Cambrian explosion. The camera eye has evolved independently at least seven times – in mammals (e.g. humans), cephalopods (e.g. squid and octopus), certain annelid worms, cubozoans (a form of jellyfish), and three separate forms of snail. Wings allegedly evolved independently no less than four times: in insects, flying reptiles, birds and bats. Electrogeneration in fish has appeared independently at least six times and in each case involved the modification of muscle cells. Bioluminescence – the ability of creatures to produce their own light with chemicals – is said to have evolved independently 40 to 65 times. The whale, dolphin, extinct ichthyosaurus of the Mesozoic, and shark all look similar, yet the shark is a fish, the ichthyosaurus was an aquatic reptile, and the whale and dolphin are mammals. Other convergences include the production of silk threads by spiders, silk moths, larval caddis flies and weaver ants, sonar-like echolocation systems in microbats, toothed whales and shrews, and warm-bloodedness in birds, mammals and certain fish.

Fig. 5.8. Convergence of the camera eye in humans (vertebrate) and the octopus (cephalopod). The eyes are ‘wired’ differently: in humans light passes through the nerves on the way to the photoreceptors (retina), whereas in the octopus it does not. 1 = retina; 2 = nerve fibres; 3 = optic nerve; 4 = blind spot in humans, caused by nerve fibres passing through the retina.
Palaeobiologist Simon Conway Morris has catalogued the extraordinary variety of convergences in animals and plants. He says that the extent and importance of convergence have been consistently underestimated, and that most examples are known only to specialists. Descriptions of convergences are full of adjectives like ‘remarkable’, ‘striking’, ‘extraordinary’, ‘astonishing’ and ‘uncanny’. Morris says that ‘there is almost a feeling of unease in the similarities’, and that some biologists ‘sense the ghost of teleology looking over their shoulders’. Life, he says, ‘shows a kind of homing instinct’;6 the ubiquity of convergence ‘means that life is not only predictable at a basic level, it also has direction’.7 But he has no explanation other than the standard neo-Darwinian tale that similar forms and structures evolve because random mutations are sifted by similar selection pressures, and because there may be only a very limited number of ways of solving particular challenges (e.g. designing an eye). However, it is difficult enough to imagine how a complex organ or organism could have evolved even once by a combination of thousands of randomly generated ‘beneficial’ mutations; the idea that it could have happened more than once beggars belief. Moreover, when related species independently evolve similar physical traits they sometimes use the same genes to do so – which deals a further blow to the idea that evolution is essentially a random process.8
Numerous examples from the fossil record therefore suggest that particular evolutionary pathways are repeated: organisms with features almost identical to previous species appear again and again. Instead of thinking in terms of random mutations, it seems more reasonable to suppose that records of past features and structures are stored in some way, and that these records can be tapped into and modified during the design of later creatures.
References
Vertebrate embryos pass through a series of similar stages in early development. As Rupert Sheldrake explains:
The early stages of embryology often resemble those of numerous other species, or even families and orders. As development proceeds, the particular features of the order, family, genus and finally species tend to appear sequentially and the relatively minor differences that distinguish the individual organism from other individuals of the same species generally appear last.1
In 1866 Ernst Haeckel formulated the ‘biogenetic law’, which states that ‘ontogeny recapitulates phylogeny’, meaning that embryological development recapitulates ancestry. He argued that an organism evolves by tacking on new stages to its process of embryonic development, so that as an organism passes through embryonic development it retraces every adult stage of its evolutionary ancestors. Biologists soon discarded the idea that evolution is limited to changes added at the end of the development process, and took the view that evolution can affect all phases of development, removing developmental steps as well as adding them, so that embryology is not a strict replay of ancestry.
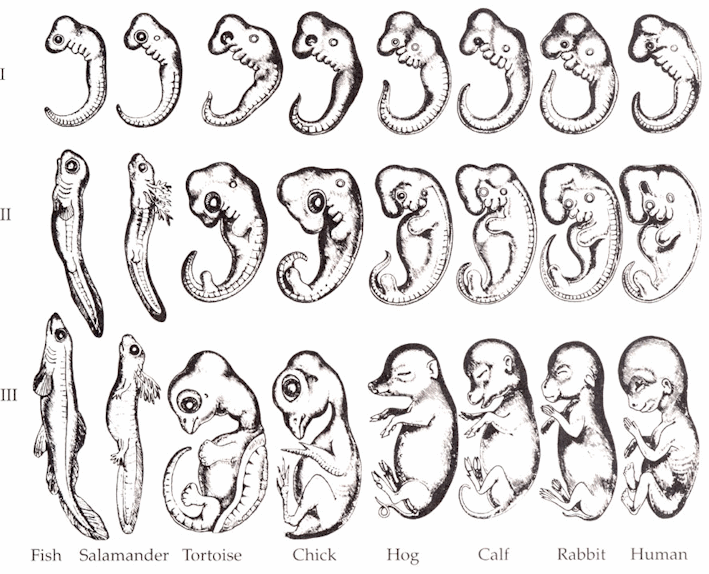
Fig. 5.9. George Romanes’s 1892 copy of Haeckel’s infamous drawings of vertebrate embryos (en.wikipedia.org). Haeckel had modified his drawings to make their ‘early’ stages appear more alike than they really are and included only a narrow range of species.2 In reality, vertebrate embryos ‘start out looking very different from each other, then they converge somewhat in appearance midway through development (Haeckel’s “first” stage) before diverging to their adult forms’.3




Fig. 5.10. Photos of (from top to bottom) a human, pig, chick, and fish embryo at similar stages of development.4
The embryo starts as a single cell, then divides into a tiny multicellular ball. A mammal embryo continues through stages resembling fish and reptiles before finishing as a fully formed mammalian youngster. Comparative embryology shows how different adult structures of many animals have the same embryonic precursors. Darwinists interpret these shared developmental features as evidence that many animals have ancestors in common; closely related animals show more similarities than more distantly related animals. For instance, at a certain stage of development, vertebrate embryos develop pharyngeal pouches resembling the gill pouches found in fish, though these features are never functioning gills, not even in embryonic fish. These features then go on to develop into very different adult structures – gills in the fish, and ear, jaw and pharynx in the mammal. This is interpreted to mean that all mammals share a common ancestor whose embryo had pharyngeal pouches.
Theosophy agrees that embryology provides information about evolutionary history, but rejects the Darwinian notion that every new type of organism arose through the continuous transformation of physical ancestors (see section 8). It should be noted that materialistic science cannot truly explain any aspect of embryonic development. For example, how does an embryo know when to stop making liver cells and to start making kidney cells? Chemical signals are believed to trigger the changes, switching certain combinations of genes on and off at just the right moments – but this raises more questions than it answers. Moreover, no known genetic mechanism explains morphogenesis or how organisms are able to retain a memory of ‘ancestral’ forms.
Another way of looking at embryological development is expressed in Von Baer’s laws, which were formulated before Haeckel’s biogenetic law. They indicate that the most generalized characters tend to appear earliest in ontogeny, followed by less generalized characters and finally the most specialized. This means that those structures that develop early in the embryo are common to many different species, whereas structures that develop late in the embryo are the ones that can be used to distinguish between species. In other words, life forms tend to begin near a common point and diverge outward, each on its own unique path, like the diverging spokes of a wheel. Von Baer was a creationist and formulated this law in opposition to evolution, but Darwinists believe that the stage of development at which two species diverge depends on how closely they are related – the assumption being that the only way they can be related is by physical descent. Theosophy postulates the existence of astral root-types, which were then developed in many different directions – not in a random fashion, but guided by nature’s instinctive intelligence.
Darwinists find further evidence of common descent in ‘vestigial organs’, which they view as the remains of what were once fully functional organs in the evolutionary ancestors of the species concerned. Some organs once labelled ‘vestigial’ have been shown to perform useful functions, e.g. the appendix, which helps fight infections and provides a safe haven for beneficial bacteria. The human coccyx (tailbone) is seen as a vestigial tail and evidence that some of our ancestors had a tail.5 The remains of a hip girdle and hind limbs in whales, and the reduced hind limbs of primitive snakes are interpreted as incomplete modifications of the structures of their ancestors. But this sort of evidence is also compatible with some kind of conscious design, since modification of certain basic structures would be more efficient than designing everything from scratch. Moreover, the lack of any substantial fossil evidence for gradual evolutionary change is consistent with the theosophical view that the preparations for new physical features and forms take place on the ethereal level.
References
Darwinists explain not only similar bodily structures but also genetic similarities in terms of common descent. But again, such similarities show nothing definite about how the organisms originated, and could just as easily be attributed to some form of conscious design.
The mainstream view used to be that the human genome contained about 100,000 protein-coding genes. However, the latest research suggests that the number may be as low as 19,000.1 These protein-coding genes make up only about 1.5% of the genome; the rest consists of noncoding DNA involved in gene regulation or other biological functions. The number of protein-coding genes in humans is not significantly larger than the number in many less complex organisms, such as the roundworm and the fruit fly.
Before DNA sequences from different species can be compared, they have to be aligned. But since the sequences typically contain repeated and/or deleted segments, it is often unclear where to start aligning them. There are computer programs for this purpose but they depend on the parameters programmed into them, and sometimes give biologically implausible results. One survey found that over 50% of evolutionary biologists and over 75% of phylogeneticists intervene manually in their sequence alignments. Data is often excluded on the grounds that not all DNA sequences carry ‘strong phylogenetic signals’, especially sequences unique to a specific lineage (known as ORFan or orphan genes). In other words, biologists commonly pick and choose their preferred alignments, and discard any unwanted sequences.2
Every species whose genome has so far been sequenced has been found to have hundreds of orphan genes. They post a huge challenge to neo-Darwinism, which predicts that genes in one taxon will typically have similar genes in closely related taxa, with the differences growing more pronounced the more distantly related the species are. There is now widespread recognition that orphan genes must have arisen spontaneously from scratch (i.e. de novo). The proportion of all protein-coding genes which originated de novo from noncoding sequences is estimated at between 10% and 30%, whereas Darwinian theory predicts that new genes should gradually emerge from other functional genes.3 The authors of a 2016 article about insect phylogeny decided to ignore orphan genes completely on the grounds that they are ‘phylogenetically uninformative’. The authors cherry-picked data from every insect they studied, discarding 40% of the sequences from the fruit fly and 80% of the sequences from the water flea, in order to produce a phylogenetic tree to their liking.4
Darwinists use differences in proteins and DNA as a ‘molecular clock’ to estimate how long ago different species diverged from a common ancestor. Each gene or protein is a separate clock, which ‘ticks’ at a different rate. For instance, it is estimated that 600 million years are required to produce a 1% difference in the histones of two different organisms, compared with 20 million years for cytochrome C, 5.8 million years for haemoglobin, and only 1.1 million years in the case of the fibrinopeptides. However, the evolutionary trees based on different classes of proteins are sometimes contradictory, and there are also major discrepancies between family trees based on comparative anatomy and those based on molecular biology.5 A study of 1070 genes in 20 different yeasts yielded 1070 different family trees. As one evolutionary biologist commented: ‘We are trying to figure out the phylogenetic relationships of 1.8 million species and can’t even sort out 20 [types of] yeast.’6 Different molecular clock studies indicate that the hypothetical common ancestor of all animals lived anywhere from about 2 billion to 274 million years ago (the latter date falls about 250 million years after the Cambrian explosion!).7 Evolution rates based on the fossil record tend to be much higher than those predicted from genetics.8
The meaning of overall DNA similarity between two organisms is a matter of debate. For instance, the genetic similarity of humans and chimpanzees has been put at 95%, 98.5%, and even 99.4%; yet humans possess self-conscious intelligence while apes do not. On the other hand, there are two species of fruit fly (Drosophila) that look alike but have only 25% of their DNA sequences in common. One study found that the snake and the crocodile (both reptiles) had only around 5% of their DNA sequences in common, whereas the crocodile and chicken had 17.5% of sequences in common – the opposite of what neo-Darwinism predicts. There are more than 3000 species of frog, all of which look superficially the same, but there is greater variation of DNA among them than between the bat and the blue whale.9 This is a further indication that far more than DNA is required to build an organism.
Evolution and Design: Contents